29 October 2024 | Tuesday | News
Picture Courtesy | Public Domain
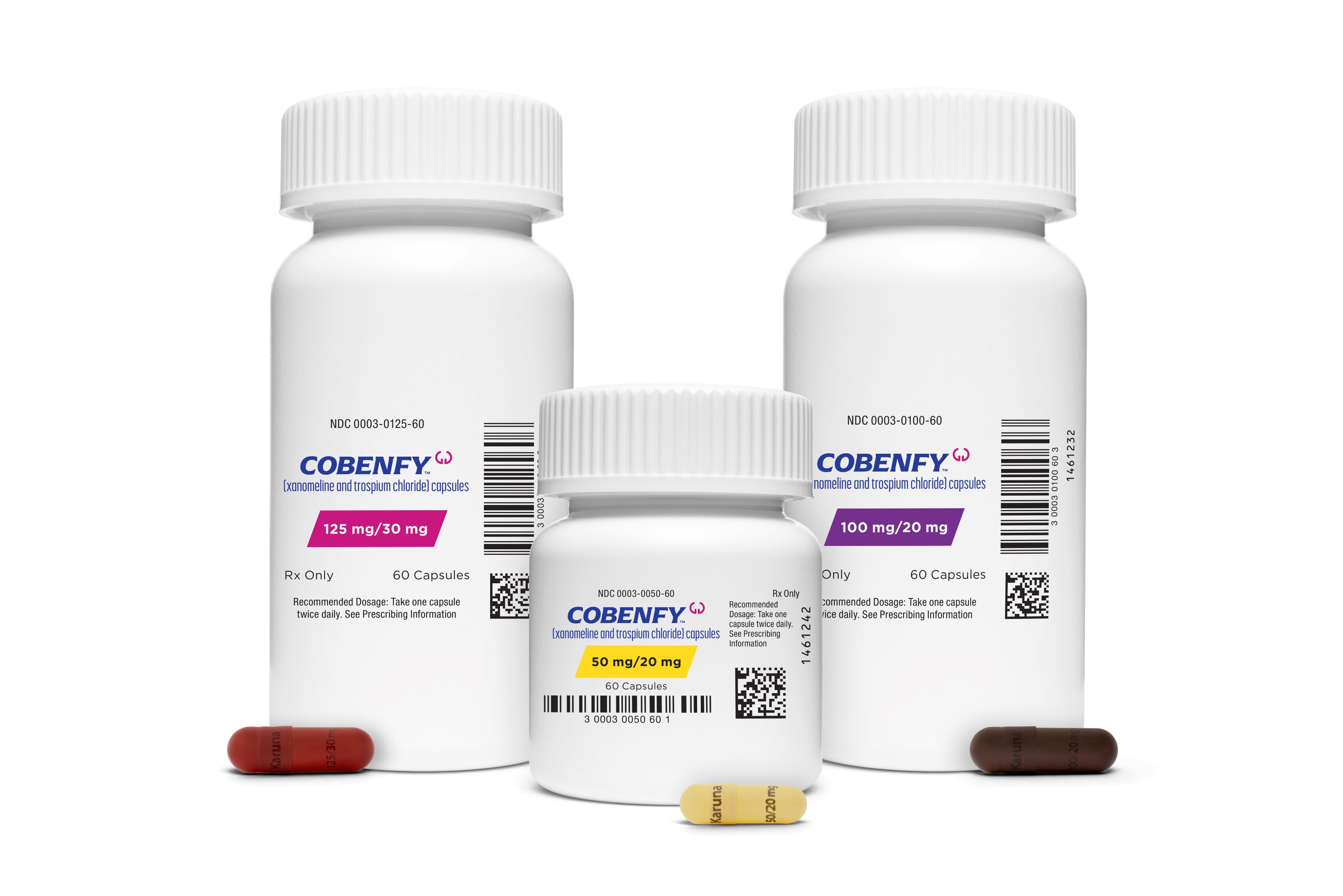
Bristol Myers Squibb (NYSE: BMY) announced that new clinical and health economics and outcomes research data (HEOR) from its neuropsychiatry portfolio evaluating COBENFY™ (xanomeline and trospium chloride) in schizophrenia in adults will be presented at Psych Congress 2024, taking place October 29 – November 2 in Boston, Massachusetts.
“Building on momentum from the recent U.S. Food and Drug Administration’s approval of COBENFY for the treatment of schizophrenia in adults, we’re pleased to be sharing additional data at Psych Congress from the EMERGENT clinical trial program that further highlights the differentiated clinical profile of COBENFY, including efficacy, safety, and patient reported outcomes of adults with schizophrenia who participated in our long-term clinical trials,” said Alyssa Johnsen, MD, PhD, senior vice president and head of clinical development, Immunology, Cardiovascular and Neuroscience, Bristol Myers Squibb. “As we continue to strengthen our neuropsychiatry portfolio, we remain committed to developing and delivering differentiated options for patients.”
Research to be presented at the meeting continues to demonstrate COBENFY as a differentiated treatment option for adults living with schizophrenia.
© 2026 Biopharma Boardroom. All Rights Reserved.