16 March 2026 | Monday | News
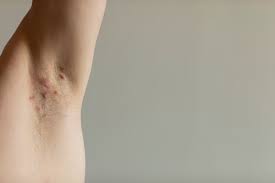
Novartis announced that Cosentyx® (secukinumab) received US Food and Drug Administration (FDA) approval for treating pediatric patients 12 years and older with moderate to severe hidradenitis suppurativa (HS), making it the only IL-17A inhibitor for this population1. The approval of a distinct biologic option for pediatric patients living with HS allows treatment to be tailored to the individual and establishes Cosentyx as a meaningful addition to the treatment landscape1-13.
“Hidradenitis suppurativa (HS) often begins in adolescence and can cause irreversible scarring and disabilities,” said Alexa B. Kimball, MD, MPH, lead investigator of the SUNSHINE and SUNRISE clinical trials in adult HS patients, President and CEO of Harvard Medical Faculty Physicians at Beth Israel Deaconess Medical Center, Boston, and Professor of Dermatology at Harvard Medical School. “The approval of Cosentyx represents an important advancement for younger HS patients who have had limited treatment options.”
HS is a chronic, systemic inflammatory skin disease that causes recurring boil-like lesions, which can rupture into painful wounds and lead to scarring7. HS affects as many as 1 in 100 people worldwide and often begins around puberty7. More than half of patients develop symptoms during adolescence, highlighting the importance of early intervention5,6.
“Hidradenitis suppurativa (HS) affects far more than skin; it impacts confidence, emotional well-being and relationships during a formative period for many pediatric patients,” said Brindley Brooks, Founder & CEO, HS Connect. “For families watching their children struggle, this FDA approval brings hope for earlier intervention.”
The distinct IL-17A mechanism provides physicians with a differentiated therapeutic option to help manage this challenging condition in younger patients, with dosing tailored to patient weight1. The use of Cosentyx in patients aged 12+ with moderate to severe HS weighing 30 kg or more is supported by well-controlled adult studies and pharmacokinetic modeling extrapolated from adult HS and psoriasis clinical trials, as well as pediatric clinical trial data from other approved indications1. The approval is also supported by dosing analysis, which predicted that weight-based dosing of Cosentyx in pediatric patients can provide similar exposure to adult HS patients1.
“With more than a decade of real-world experience across multiple autoimmune diseases, Cosentyx is a well-established treatment option that many physicians trust,” said Victor Bultó, President, Novartis US. “Yet for young people living with moderate to severe hidradenitis suppurativa (HS), treatment options have remained limited for far too long. Expanding Cosentyx to this population addresses a critical gap in care and underscores our focus on advancing solutions where we can make the greatest impact on outcomes.”
© 2026 Biopharma Boardroom. All Rights Reserved.