17 April 2026 | Friday | News
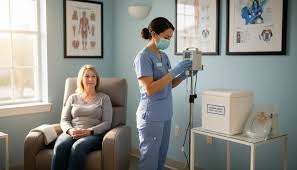
Nkarta, Inc. (Nasdaq: NKTX), a clinical-stage biotechnology company developing engineered natural killer (NK) cell therapies to treat autoimmune diseases, announced that it has reached agreement with the U.S. Food and Drug Administration (FDA) on key changes to the ongoing Ntrust-1 and Ntrust-2 clinical trials. The protocol amendments are designed to alleviate the need for overnight stays and reduce the overall burden on patients, enabling outpatient administration of NKX019 – an investigational CAR-NK cell therapy – by community research centers and community rheumatologists.
“We are pleased to reach agreement with the FDA on several protocol enhancements that will meaningfully improve the patient and trial site experience while supporting continued advancement of our NKX019 clinical program,” said Paul J. Hastings, Chief Executive Officer of Nkarta. “Reaching agreement with the FDA on outpatient dosing – reducing the need for patient monitoring from 24 hours to 2 hours – will reduce patient burden and expand access. It will also allow us to partner with community rheumatology centers, better positioning us to execute efficiently across our clinical program while expanding access to next-generation innovation to people from all walks of life, putting patients first.”
“Our agreement with the FDA expands who we can treat – and how – in clinical trials. When the amended protocol completes IRB review and is finalized, we can begin enrolling patients with rheumatoid arthritis in Ntrust-2. We’re also gaining the flexibility to re-dose participants in both studies, if needed, as an option to help optimize treatment responses in individual patients.”
NKX019 Clinical Program Progress and Upcoming Milestones
Following submission of the final amendments to FDA and IRB approvals:
© 2026 Biopharma Boardroom. All Rights Reserved.